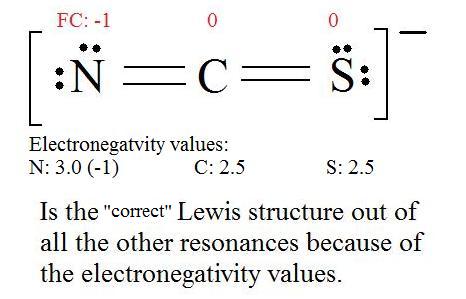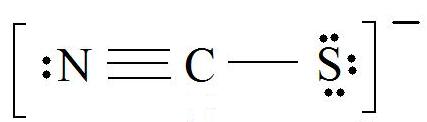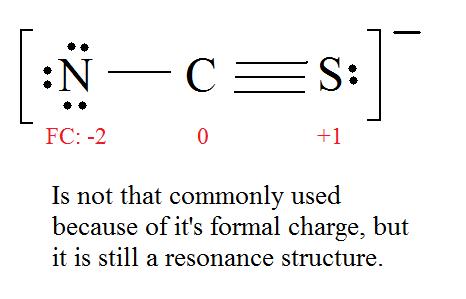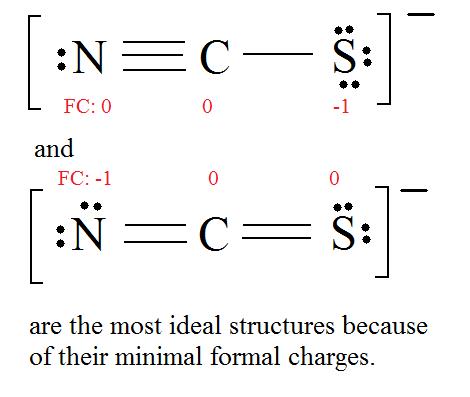PPT - formal charge : the charge a bonded atom would have if all the atoms had the same PowerPoint Presentation - ID:4506277

For thiocyanate ion SCN , three resonating structures are possible with the electron dot method as shown below:abCThe decreasing order of % contribution to the resonance hybrid is:A. Cannot be predicted B.
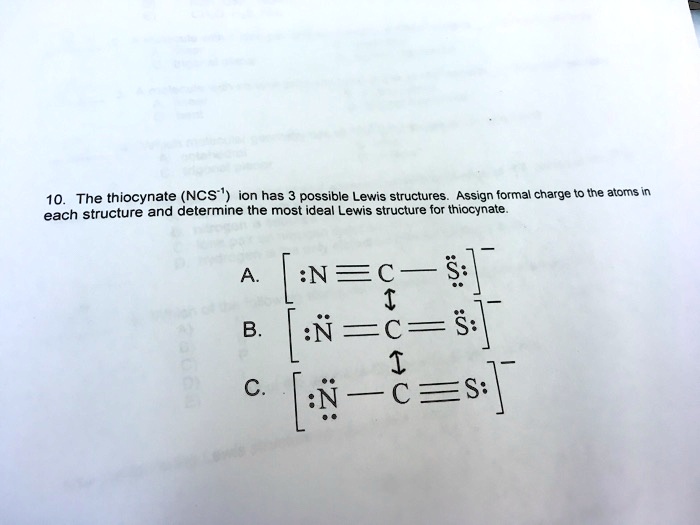
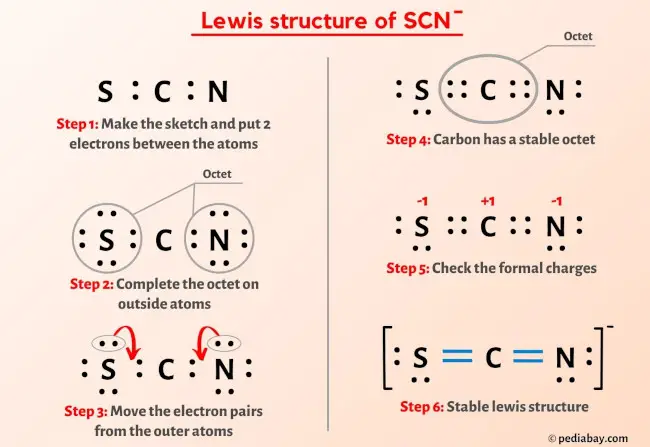
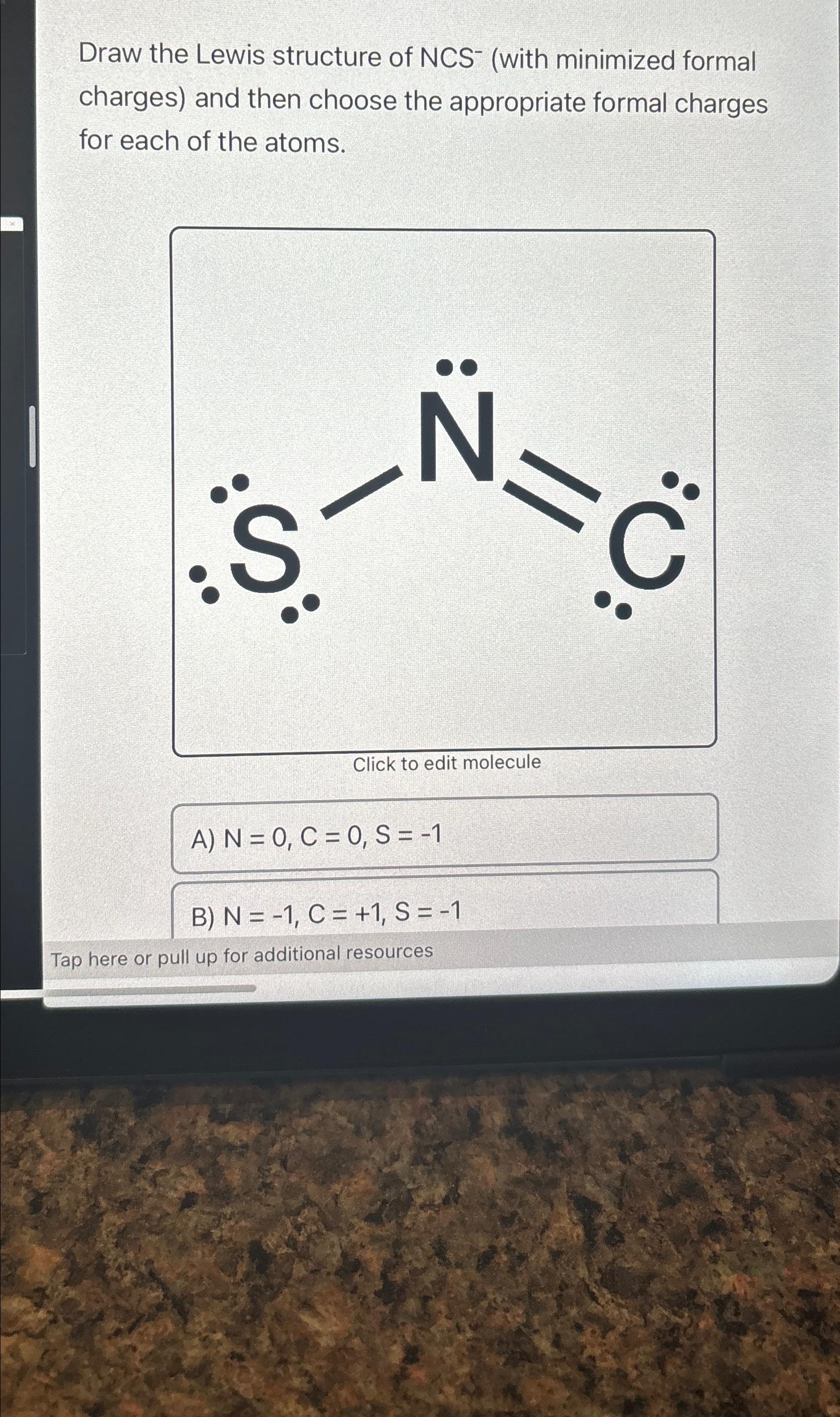
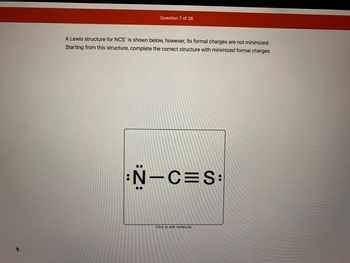
SOLVED: The thiocyanate (NCS-) ion has multiple possible Lewis structures. Assign formal charges to the atoms in each structure and determine the most ideal Lewis structure for thiocyanate. N=C=S-